What Benefit Does Having a Cell Wall Made of Cellulose Provide to Land Plants
Introduction
Plant cell walls are multifunctional polysaccharide-rich fibrous composites in which polymers collaborate to form load-bearing structures embedded in a polysaccharide matrix (Bacic et al., 1988; Fry, 2004). Cells in the growing parts of plants are bound by "master walls" in which the load begetting function is performed primarily by cellulose microfibrils. Models of the establish cell wall typically draw the microfibrils cross-linked with hemicelluloses, including mannans, xylans, mixed-linkage glucans (MLG), and xyloglucans. This network is and then further embedded in a matrix of pectic polysaccharides including homogalacturonan (HG), rhamnogalacturonan-I (RG-I), rhamnogalacturonan-2 (RG-Two), and xylogalacturonan (Fry, 2004; Mohnen, 2008; Caffall and Mohnen, 2009; Harholt et al., 2010). However, this conventional description of chief walls that emphasizes tethering glycans as indispensible "load-bearing" structures may need revising every bit discussed in Scheller and Ulvskov (2010). Primary jail cell walls establish the foundations for cell shape and resist the tensile forces exerted by turgor force per unit area. They must too be capable of controlled expansion to enable cell growth. In non-growing constitute tissues, some cells are typically surrounded by "secondary walls" whose primary role is to resist compressive force and since cell expansion is non required, these walls are frequently reinforced with lignin (Hepler et al., 1970; Carpita and Gibeaut, 1993; Boerjan et al., 2003; Cosgrove, 2005). Although these descriptions serve to describe many plant jail cell walls in wide terms, they are generalizations and are primarily based on investigations of the cell walls of flowering plants. However, cell walls brandish remarkable diversity at many levels and their constituent polysaccharides differ in fine structure, relative abundance, and molecular associations (Burton et al., 2010). The vast complication and heterogeneity of cell wall glycomes is the product of the cooperative activities of prodigious numbers of biosynthetic enzymes. It is clear from genome sequencing that hundreds of glycosyltransferases (GTs) catalyze the formation of glycosidic linkages in polysaccharides –- more than than l for the pectic polymers alone (Scheible and Pauly, 2004; Mohnen, 2008; Yin et al., 2010; Dhugga, 2012). Most GTs act in the Golgi apparatus and their products are transported to jail cell walls in secretory vesicles. In contrast, cellulose- and callose synthases, and possibly the "D" class of cellulose synthase-similar GTs, are embedded in the plasma membrane and their products are extruded directly into cell walls (Endler and Persson, 2011; Park et al., 2011). The big numbers of GT-encoding genes and their varied temporal and spatial expression profiles produce vast possibilities for cell wall variability. Further heterogeneity is generated past the availability of a wide range of activated sugar donors (Feingold and Avigad, 1980), methylation and acetylation, different enantiomer and the variety and number of possible glycosyl linkages besides as in muro modification of polysaccharides, e.g., by esterification/deesterification of pectins and transglycosylation between certain hemicelluloses (Fry et al., 2008; Burton et al., 2010). Collectively, these dynamic processes enable plants to generate cell walls that are exquisitely suited to prevailing functional requirements and that can respond to biotic and abiotic stresses equally well as developmental cues (Sarkar et al., 2009; Sørensen et al., 2010).
Why Study Cell Wall Variety?
The report of jail cell wall glycomes across the establish kingdom is of import for developing our understanding of cell wall structures and functions, for understanding cell wall and institute evolution, and for optimizing the utilization of the largest source of biomass on globe. Plants emerged onto land around 470 million years ago and have since colonized a large proportion of the World's surface (Kenrick and Crane, 1997; Waters, 2003; Niklas and Kutschera, 2010). The transition to land was a pivotal event in the history of life which resulted in the germination of new habitats and ecosystems and had profound furnishings on atmospheric chemistry. Cell walls have played significant roles in these epochal evolutionary events but our electric current understanding of many aspects of cell wall structures and their evolution is express (Niklas, 2004; Popper and Tuohy, 2010; Sørensen et al., 2010). Improving our understanding will contribute to a wider agreement of constitute evolution and phylogenetic relationships and may provide noesis about past environments and insight into how plants might answer to predicted climate alter scenarios.
The study of jail cell wall evolution is based largely upon the surveying of jail cell wall diversity (Popper, 2008; Sørensen et al., 2010; Popper et al., 2011). Simply by doing this is it possible to correlate changes in plant physiology, morphology or addiction with corresponding innovations in jail cell wall biology. A study of prison cell wall diversity across the found kingdom also has other benefits. Prison cell wall polysaccharides are an immensely valuable renewable bio-resource and accept numerous industrial applications. Timber, fibers, paper, functional ingredients (e.g., pectins from flowering plants and alginates and carrageenans from algae), and nutraceuticals, and first and second generation biofuels are predominantly prison cell wall-based (Bacic et al., 1988; Willats et al., 2006; Pauly and Keegstra, 2010). In contrast to nucleotide sequences and proteins, polysaccharides cannot readily be synthesized and so must be sourced from nature. Currently nosotros apply just a minute fraction of the global cell wall glycome and a comprehensive inventory of bachelor polysaccharides may reveal valuable new molecules and materials with novel uses. The analysis of various jail cell wall compositions and architectures might also provide inspiration for current efforts aimed at the targeted modification of cell walls, notably for energy crops. Withal, surveying of cell walls across the plant kingdom is a daunting undertaking which equally described below, entails many significant challenges and requires a multi-disciplinary approach. This is in large office considering polysaccharides are non directly encoded by genome sequence; multiple enzymes are required to synthesize the activated sugar residues, linkages and many wall components undergo extensive modifications including methylation, esterification/deesterification, and acetylation as well equally the add-on of unmarried or blocks of carbohydrate residues.
Challenges in Surveying Cell Wall Multifariousness
The specific genes and enzymes that lead to synthesis of specific cell wall components has yet to exist fully elucidated. Furthermore, in the majority of cases several enzymes are required to synthesize a specific jail cell wall component which may additionally undergo subsequent modification in muro. Consequently we are not yet at the stage where information technology is possible to determine cell wall composition and diversity via a comparative genomics arroyo and much of the knowledge so far gleaned has relied on polymer analysis. One primal difficulty associated with this is that polysaccharides are not acquiescent to facile sequencing. Their structures can be determined by several well established chemical methods which take been developed and applied to cell wall studies over the last l or so years. Each method has both limitations and merits but they may exist applied in concert to reveal and determine jail cell wall complication and multifariousness. Few of the methods developed so far are amenable to high throughput screening, so wide surveys have to rely on partial label initially. Fourier Transform Infra-Ruby-red Spectroscopy (FTIR) requiring fiddling sample preparation tin can be high throughput and is useful for determining differences in cell wall limerick across samples but is rarely effective for precise compositional assay as it does not yield sequence information (Mouille et al., 2003). Recently, methods based on carbohydrate microarrays probed with monoclonal antibodies (mAbs) and saccharide-binding modules (CBMs) with specificities for jail cell wall polysaccharide epitopes have been developed. This technology can enable analysis of the occurrence of 100 epitopes in 2–three days and can reveal much virtually prison cell wall composition despite some limitations largely derived from the current, although increasing, availability of characterized mAbs and CBMs (Moller et al., 2007; Sørensen et al., 2009; Pattathil et al., 2010). Awarding of FTIR and saccharide microarrays tin exist used to indicate cell walls that have a composition that may merit farther and detailed analysis because they announced to be significantly different from characterized cell walls. Thus, utilize of these methods facilitates the evolution of hypotheses regarding prison cell wall composition that can be further explored by more detailed analysis of a subset of the sample prepare. Polysaccharide gel electrophoresis (PACE) (Goubet et al., 2002), Oligosaccharide mass profiling (OLIMP; Lerouxel et al., 2002), paper chromatography, and sparse layer chromatography (Fry, 2001) and related approaches are powerful tools for the next level operating on a subset of the original sample set. Glycosyl linkage analyses and NMR make upwardly the concluding tier as these methods are time consuming and for NMR also quite insensitive. These techniques are thus unsuitable for wide calibration sampling only are often indispensible for in-depth analysis of selected samples. Whatever method of assay is chosen, sampling will e'er pose significant challenges. Given that information technology is not feasible to sample the walls of every extant plant then what plants and what organs or tissues should exist chosen and is there sufficient tissue bachelor? It seems reasonable to select species that are representative of taxonomic groups or morphotypes, just such prioritizations are not e'er straightforward. One time plants are chosen then one is faced with the further difficulty of properly sampling the individual prison cell walls inside that plant. One option would exist to simply homogenize the whole found and extract every bit many jail cell wall polysaccharides every bit possible. This is feasible for say, small seedlings and microalgae, but not for woody species and copse. Clearly then, certain tissues, organs or developmental stages need to exist selected – but on what ground? Equivalence can also be problematic because plants differ in the organs and tissues they have. Some have flowers and leaves, some do not. Additionally, contradistinct growth conditions may touch on the expression and structure of prison cell wall components within the same species (Iraki et al., 1989a,b,c). Such considerations are important because nosotros know that some cell wall components can be very selectively distributed throughout a found (encounter section Fine mapping of cell wall multifariousness and heterogeneity at the cellular and subcellular levels) and tin can easily be missed. Interpretation of various analyses is a further important claiming. Considerable caution is required considering the near impossibility of truly inclusive sampling. If positively identified by multiple methods and then a particular polysaccharide can exist regarded as "nowadays," but unless all parts of a constitute have been sampled (including all developmental stages) then failure to identify a particular polysaccharide should be interpreted as the presence of that polysaccharide being "unknown" rather than "absent-minded." When investigating cell wall evolution it is also important to consider polysaccharides that may occur at such low levels that they may be regarded as functionally unimportant in other studies. For example, although glycosyl linkage and ICP-MS information suggest RG-Ii or RG-Ii-like oligosaccharide occurs at very low levels in avascular bryophytes, less than ane% of that in angiosperms (Matsunaga et al., 2004), its presence in these plants would nevertheless be significant in terms of the evolution of underlying biosynthetic mechanisms.
A Multi-Level Arroyo Towards Understanding Jail cell Wall Variety and Evolution
The authors have adopted a multi-level strategy for mapping cell wall polysaccharide and genetic diversity in society to gain insight into underlying evolutionary mechanisms (Figure 1A). The first level consists of primary screens for cell wall polysaccharides based on carbohydrate microarrays probed with mAbs and CBMs (Moller et al., 2007). This level is limited by the availability of characterized mAbs and CBMs and their ability to recognize the numerous epitopes which occur in the various found prison cell wall components. Plain the ideal would be to have as much coverage as possible of all the epitopes that exist within cell wall components. However, this is non the electric current situation and at that place are some notable wall components, such equally RG-2, for which an effective mAb has withal to exist generated. In parallel to saccharide microarrays, genomes and transcriptomes are mined to identify jail cell wall-related GTs. The 2nd level of assay seeks to obtain more than detailed data nearly sure polysaccharides and genes from subsets of plants. These analyses are performed using established methods for polysaccharide assay and gene cloning and sequencing. A tertiary level is aimed at obtaining definitive data and the functions of genes, poly peptide, and polysaccharides. In some cases genes are expressed and biochemical activities of GTs adamant. Figure 1B shows some preliminary data from main screens of polysaccharides and cell wall-related genes. The combined analysis of data sets tin can provide insight into the timing and machinery of certain evolutionary events. For example, Xue and Fry (2012) have suggested that MLGs are restricted to horsetails based on the results obtained when diverse monilophyte cell walls were treated with lichenase, an enzyme that specifically fragments these (1→three)(ane→4)-β-D-glucans. In contrast, nosotros have obtained testify for MLGs in Selaginella moellendorffii and selected Charophycean green algae (CGA) using microarray-based polysaccharide screening and lichenase treatments (Harholt et al., 2012; Fangel and Willats, unpublished). Since a genome sequence is available for South. moellendorffii information technology is possible to establish with confidence that this plant does not comprise orthologs of the CSLF and CSLH genes (Harholt et al., 2012) that are implicated in the synthesis of (i→3)(1→4)-β-D-glucan in Poales species (Burton et al., 2006; Doblin et al., 2009). These information provide proficient testify that (1→3)(1→4)-β-D-glucan has evolved at least twice by convergent evolution.
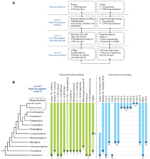
Figure 1. (A) A multi-level approach is required to assess the occurrence of prison cell wall polysaccharides and related genes throughout the institute kingdom. (B) Selected data from primary screens of polysaccharide occurrence in diverse plant species and results from mining genomes and transcriptomes for cell wall-related GTs. The dots at the bottom of the bars indicate the most basal plant group in which that particular polysaccharide or gene sequence has so far been identified. The confined indicate that it is causeless in most cases, once evolved, a factor or polysaccharide persists throughout evolution and is present in later diverging species. Notwithstanding, this is by no means universal. For example, the dashed bar for (one→3)(1→4)-β-D-glucan indicates that the occurrence of this polysaccharide is intermittent throughout the plant kingdom and has arisen past convergent development more than once. In some cases the presence of jail cell wall components is equivocal. For example, lignin has been tentatively identified in certain Charophycean light-green algae but the most basal group in which it has been definitively identified is the Lycopodiophyta. A clade of putative bequeathed CSLs that are common ancestors to the CSLAsouth and CSLCs has been identified in certain Chlorophyte algae by Yin et al. (2009). These genes share homology with both CSLAs and CSLCsouth but appear to differ sufficiently to warrant a new family name, which we take assigned CSLKs, consistent with the existing alphabetical sequence of the existing CSL family names. (1) Tsekos (1999); (2) Scherp et al. (2001); (3) Mackie and Sto (1968); (4) Domozych et al. (2009); (5) Sørensen et al. (2011); (half dozen) Matsunaga et al. (2004); (7) Estevez et al. (2009); (eight) Weng et al. (2008); (9) Roberts et al. (2002); (10) Abercrombie et al. (2011); (eleven) Yin et al. (2009); (12) Yin et al. (2010); (13) Egelund et al. (2007), Velasquez et al. (2011); (14) Qu et al. (2004), Harholt et al. (2012). TC, terminal complex.
Genome and Transcriptome Mining for Cell Wall-Related Glycosyl Transferases
"At least ane GT for each glycosidic linkage" is axiomatic for our bioinformatic analysis of cell wall biogenesis. Rare dual role GTs are known from animals and plants, with CslA as an example of a GT that can utilize ii different Gdp-sugars as substrate. Accepting the axiom of one GT per glycosidic linkage should permit inference of the minimum number of GTs involved in cell wall biosynthesis. However, this is not possible for ii reasons. Firstly, the evolution of flowering plants was accompanied by very substantial factor duplication. Differentiation of complex tissues in angiosperms calling for separate regulation in space and time of the same catalytic activity is a probable contributing factor. Mitchell et al. (2007) combined this line of thinking with expression assay and proposed that sure clades of family GT61 should comprise genes involved in synthesis of a polymer of detail importance in grasses, a prediction that was recently proven right (Anders et al., 2012). The large repertoire of GTs of the moss Physcomitrella patens, despite being non-vascular (then having not-lignified tissues only) and also having diverged prior to the gene duplication events associated with the development of flowering plants, cautions us non to generalize this principle excessively. The second reason why the number of GTs cannot be inferred from the number of different linkages is that the biosynthesis of some polysaccharides has turned out to be much more complicated than anticipated from the polysaccharide structure; xylan biosynthesis, reviewed past Scheller and Ulvskov (2010), provides a hit example. In dicots, GTs from two different families (GT43 and GT47) are implicated in synthesizing the xylan backbone totaling 8 GTs for the synthesis of one linkage. Pectin biosynthesis is predicted to require at to the lowest degree 67 enzymes, GTs plus methyl- and acetyl transferases (Mohnen, 2008). Too few of these have been identified and then far to guess whether Nature's approach to pectin biosynthesis is lean, or expansive as with xylan. These limitations still, it has proven fruitful to mine genomes for their GT repertoire. This is commonly done using the CAZy database equally a foundation, See Box 1. The CAZy database is the about extensive database of GTs and contains GTs from all iii kingdoms. Past using a global approach including the whole CAZy database in the screen, more remote orthologies can be discovered, exemplified by the discovery of a mannosylglycerate synthase of GT78 in S. moellendorffii (Scheller et al., 2010). Simply CAZy is not complete and GTs may be establish outside CAZy and certain activities are hard to account for within the limit of the present CAZy database (Hansen et al., 2012). The CAZy driven approach has been applied to poplar (Geisler-Lee et al., 2006) and most recently to Brachypodium distachyon (Vogel et al., 2010) and S. moellendorffii (Banks et al., 2011) also leading to a CAZy-based naming convention for putative GTs that tin be assigned to a factor family unit just not to a office.
BOX i Text Box entitled CAZy.
The CAZy database, www.cazy.org, describes the families of structurally related catalytic and carbohydrate-binding modules (or functional domains) of enzymes that degrade, alter, or create glycosidic bonds (Cantarel et al., 2009). GTs are classified to 94 families (and growing). The classification is partly based on sequence similarity, partly on 3D information of poly peptide structure. The 94 families can be grouped into a small number of GT-folds creating a college level in the bureaucracy formalized as clans for glycoside hydrolases but not even so for GTs. About families comprise several activities so assigning a GT to a family unit is rarely sufficient for deducing the precise catalytic action of the GT. GTs may, however, with good precision exist predicted to be inverting or retaining based on their CAZy family, where retaining refers to the situation where the transferred monosaccharide ends up in the aforementioned anomeric conformation equally in the donor substrate and conversely for inverting GTs.
Applying the CAZy-based classification of putative GTs across large phylogenetic distances can yield evolutionary relevant data every bit exemplified by the case for convergent evolution of MLG. CAZy may too be employed as a support for gene discovery efforts every bit Egelund et al. (2007) did using homology between Chlamydomonas and Arabidopsis genes in clade A of family unit GT77 to infer the part of the Reduced Balance Arabinose (RRA) genes equally encoding putative extensin arabinosyl transferases. This annotation is not proven but was corroborated by Velasquez et al. (2011). Extensin along with mannan are to the authors' noesis the only known shared cell wall components between Chlorophycean green algae and Arabidopsis (Figure ane; Estevez et al., 2009).
No CGA has yet been sequenced which is unfortunate given that prison cell wall assay strongly suggests that the mutual ancestor of all plants, with xyloglucan and the pectic polymers typical of vascular plants, was a CGA (Figure 1). A number of EST datasets (Timme and Delwiche, 2010; Wodniok et al., 2011; Timme et al., 2012) are bachelor and may be subjected to the same CAZy-based analysis equally full genomes, albeit less safely. Our unpublished observations of this nature, in combination with recent phylogenetic analyses based on genomic data, lead us to propose that genera similar Penium and Coleochaete represent the earliest versions of a higher plant prison cell wall while species similar Chara, which superficially looks more avant-garde, has diverged substantially from the main evolutionary path leading to terrestrial plants and hence is a less attractive model for tracing the evolutionary history of the plants cell wall (Timme et al., 2012).
Fine Mapping of Jail cell Wall Diversity and Heterogeneity at the Cellular and Subcellular Levels
Cellulose is widely distributed in most cell walls but information technology is clear that the known structural diversity of the polysaccharides of the hemicellulose and pectic groups is regulated both taxonomically and also in relation to jail cell blazon and cell wall microstructures within tissues. The use of mAbs and CBMs for in situ analyses of cell walls remains a key approach to determine wall molecular architectures and their heterogeneities (Burton et al., 2010; Pattathil et al., 2010; Lee et al., 2011). The use of the same molecular probes in glycomic analysis is a very useful and complementary activity in which specific oligosaccharide structural features can be studied widely in terms of occurrence and biochemistry in addition to cellular locations.
It is a meaning betoken that although we know some broad cellular occurrences of specific not-cellulosic polysaccharides – such equally the abundance of xylan in secondary cell walls of dicotyledons, of HG in primary cell walls and (an instance from the subcellular level) the absence of pectic galactan from pit fields, we do not have a skilful understanding of the cellular distributions of all polymers for near plant cell types. Even for Arabidopsis a systematic in situ assessment of the major polymers in the cell walls of the major organs has non yet been achieved. Some can be predicted – such every bit those indicated above but the distributions of RG-I and other pectic epitopes or hemicelluloses cannot all the same be predicted with certainty. In situ analyses of cell wall structures are made more than complex but also more revealing in that as probe sets for specific polysaccharides are extended more prison cell wall diversity and heterogeneity can be uncovered. For case, this is the instance for pectic HG (Willats et al., 2001; Parre and Geitmann, 2005; Wolf et al., 2009) and also for pectic arabinan (Verhertbruggen et al., 2009) where a mAb for linear arabinan detects arabinan substructures in restricted intercellular regions of parenchyma as shown in Figure 2. This likewise applies to xylan structures in the same parenchyma (Hervé, 2009). What is the functional basis for polysaccharide fine construction of both xylans and arabinans beingness so intimately spatially regulated in relation to factors such as jail cell adhesion (Figure two)? Is this to be a prototype for non-cellulosic wall polymers for which we currently accept ane or a express number of probes such as pectic galactan and xyloglucan? The adjacent few years will see more detailed systematic assessments of molecular architectures. This will be in conjunction with enzymatic and/or chemic pre-treatments that are in some cases required to optimize polysaccharide detection and in the example of polysaccharide masking (in which 1 polymer class blocks admission to another polymer class) indicates important features of cell wall architectures reflecting poly peptide admission (Vreeland et al., 1984; Marcus et al., 2010; Davies et al., 2012).
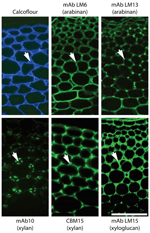
FIGURE 2. Architectural heterogeneity in main cell walls. Equivalent transverse sections of the cortical region of tobacco stems with immunofluorescence imaging of two arabinan epitopes (mAbs LM6 and LM13), two xylan epitopes (mAb LM10 and CBM15) and ane xyloglucan epitope (mAb LM15). Arrows signal intercellular matrix shared by side by side cells. Xylan and xyloglucan epitopes are shown afterward removal of pectic homogalacturonan. Pectic arabinan, xylan, and xyloglucan structural features all brandish spatial heterogeneity in relation to prison cell wall thickening and intercellular regions. mAb, monoclonal antibody; CBM, carbohydrate-binding module. Scale bar = 100 μm.
Clearly we confront many challenges in understanding cell wall development not least of which include sufficient sampling followed by appropriate synthesis and interpretation of large data sets including various information such as gene and protein sequence data as well as sugar linkages and epitope distributions. However, as cell and tissue molecular architectures are documented another major issue that is brought into focus is the function of individual wall components and of the entire prison cell walls, which tin can vary enormously with respect to quantitative and qualitative limerick. Why exercise some cell walls have xyloglucan, xylan, and mannan hemicelluloses in distinct spatial distributions as for example in the extensively studied tobacco stem system as shown in Figure 2? How are these heterogeneities integrated into a functional whole in terms of wall properties and functions? In vitro analyses of composites formed from cellulose, pectins, and xyloglucans take yielded invaluable data about the properties of some wall components (Chanliaud et al., 2002). However, the structural and compositional complexity of naturally occurring prison cell walls means that an in vitro arroyo cannot reasonably be applied to investigate the functional properties of the full diversity of extant walls. In vivo methods of investigating wall biomechanics, at the tissue and lower levels, take been developed (Spatz et al., 1998; Burget, 2006) facilitating an improved understanding of how walls are assembled and the fine detail of wall domains and their nanomechanical properties are likely to emerge within the next few years as detailed in situ analyses are combined with genetic and enzymatic interventions. Integrating this knowledge will be a major challenge and is an exciting borderland for cell wall biology.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of whatsoever commercial or financial relationships that could be construed equally a potential conflict of involvement.
References
Abercrombie, J. Yard., O'Meara, B. C., Moffatt, A. R., and Williams, J. H. (2011). Developmental evolution of flowering plant pollen tube prison cell walls: callose synthase (CalS) gene expression patterns. EvoDevo 2, 14.
Pubmed Abstract | Pubmed Total Text | CrossRef Full Text
Anders, Due north., Wilkinson, M. D., Lovegrove, A., Freeman, J., Tryfona, T., Pellny, T. K., Weimar, T., Mortimer, J. C., Stott, Grand., Bakery, J. M., Defoin-Platel, M., Shewry, P. R., Dupree, P., and Mitchell, R. A. C. (2012). Glycosyl transferases in family 61 mediate arabinofuranosyl transfer onto xylan in grasses. Proc. Natl. Acad. Sci. U.S.A. 17, 989–993.
Pubmed Abstract | Pubmed Total Text | CrossRef Full Text
Bacic, A., Harris, A. J., and Rock, B. A. (1988). "Structure and function of institute jail cell walls," in The Biochemistry of Plants, ed J. Preiss (New York: Bookish Press), 297–371.
Banks, J. A., Nishiyama, T., Hasebe, M., Bowman, J. L., Gribskov, M., dePamphilis, C., Albert, V. A., Aono, N., Aoyama, T., Ambrose, B. A., Ashton, N. W., Axtell, M. J., Barker, E., Barker, M. Due south., Bennetzen, J. L., Bonawitz, Due north. D., Chapple, C., Cheng, C., Correa, L. K., Dacre, Yard., DeBarry, J., Dreyer, I., Elias, One thousand., Engstrom, E. M., Estelle, One thousand., Feng, L., Finet, C., Floyd, S. One thousand., Frommer, W. B., Fujita, T., Gramzow, L., Gutensohn, M., Harholt, J., Hattori, M., Heyl, A., Hirai, T., Hiwatashi, Y., Ishikawa, 1000., Iwata, K., Karol, M. G., Koehler, B., Kolukisaoglu, U., Kubo, M., Kurata, T., Lalonde, S., Li, K., Li, Y., Litt, A., Lyons, E., Manning, G., Maruyama, T., Michael, T. P., Mikami, K., Miyazaki, S., Morinaga, S., Murata, T., Mueller-Roeber, B., Nelson, D. R., Obara, Yard., Oguri, Y., Olmstead, R. G., Onodera, N., Petersen, B. L., Pils, B., Prigge, M., Rensing, S. A., Riaño-Pachón, D. Chiliad., Roberts, A. Due west., Sato, Y., Scheller, H. V., Schulz, B., Schulz, C., Shakirov, Due east. V., Shibagaki, N., Shinohara, N., Shippen, D. E., Sørensen, I., Sotooka, R., Sugimoto, N., Sugita, M., Sumikawa, N., Tanurdzic, G., Theissen, Thou., Ulvskov, P., Wakazuki, Southward., Weng, J. K., Willats, W. Westward., Wipf, D., Wolf, P. G., Yang, L., Zimmer, A. D., Zhu, Q., Mitros, T., Hellsten, U., Loqué, D., Otillar, R., Salamov, A., Schmutz, J., Shapiro, H., Lindquist, East., Lucas, S., Rokhsar, D., and Grigoriev, I. Five. (2011). The Selaginella genome identifies genetic changes associated with the evolution of vascular plants. Science 332, 960–963.
Pubmed Abstruse | Pubmed Full Text | CrossRef Full Text
Burton, R. A., Wilson, S. One thousand., Hrmova, M., Harvey, A. J., Shirley, N. J., Medhurst, A., Stone, B. A., Newbigin, E. J., Bacic, A., and Fincher, M. B. (2006). Cellulose synthase-similar CslF genes mediate the synthesis of jail cell wall (1,3;1,4)-β-D-glucans. Science 311, 1940–1942.
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Cantarel, B. L., Coutinho, P. M., Rancurel, C., Bernard, T., Lombard, V., and, Henrissat, B. (2009). The Sugar-Active EnZymes database (CAZy): an adept resource for glycogenomics. Nucleic Acids Res. 37, 233–238.
Pubmed Abstruse | Pubmed Full Text | CrossRef Full Text
Carpita, N. C., and Gibeaut, D. One thousand. (1993). Structural models of primary cell walls in flowering plants: consistency of molecular structure with the physical properties of the walls during growth. Institute J. 3, 1–30.
Pubmed Abstract | Pubmed Total Text | CrossRef Full Text
Davies, L. J., Lilley, C. J., Knox, J. P., and Urwin, P. Eastward. (2012). Syncytia formed by adult female Heterodera schachtii on Arabidopsis thaliana roots have a singled-out cell wall molecular compages. New Phytol. (in press).
Doblin, M. S., Pettolino, F. A., Wilson, S. Thousand., Campbell, R., Burton, R. A., Fincher, K. B., Newbigin, E., and Bacic, A. (2009). A barley cellulose synthase-like CSLH gene mediates (1,3;1,4)-β-D-glucan synthesis in transgenic Arabidopsis. Proc. Natl. Acad. Sci. UsA. 106, 5996–6001.
Pubmed Abstract | Pubmed Total Text | CrossRef Full Text
Domozych, D. S., Sørensen, I., and Willats, Due west. G. T. (2009). The distribution of prison cell wall polymers during antheridium development and spermatogenesis in the Charophycean green alga, Chara corallina. Ann. Bot. 104, 1045–1056.
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Egelund, J., Obel, Northward., Ulvskov, P., Geshi, Due north., Pauly, Grand., Bacic, A., and Petersen, B. L. (2007). Molecular characterization of two Arabidopsis thaliana glycosyltransferase mutants, rra1 and rra2, which have a reduced remainder arabinose content in a polymer tightly associated with the cellulosic wall residuum. Plant Mol. Biol. 64, 439–451.
Pubmed Abstruse | Pubmed Total Text | CrossRef Full Text
Estevez, J. M., Fernández, P. V., Kasulin, L., Dupree, P., and Ciancia, One thousand. (2009). Chemical and in situ label of macromolecular components of the cell walls from the dark-green seaweed Codium fragile. Glycobiology nineteen, 212–228.
Pubmed Abstract | Pubmed Total Text | CrossRef Total Text
Feingold, D. South., and Avigad, Thousand. (1980). "Sugar nucleotide transformations in plants," in The Biochemistry of Plants: A Comprehensive Treatise, eds P. K. Stumpf and E. E. Conn (New York: Academic Press), 101–170.
Fry, S. C. (2001). The Growing Institute Cell Wall: Chemic and Metabolic Analysis, Reprint edition. New Bailiwick of jersey: The Blackburn Printing.
Fry, S. C. (2004). Chief cell wall metabolism: tracking the careers of wall polymers in living plant cells. New Phytol. 161, 641–675.
CrossRef Total Text
Fry, S. C., Mohler, 1000. E., Nesselrode, B. H. W. A., and Franková, L. (2008). Mixed-linkage β-glucan: xyloglucan endotransglucosylase, a novel wall-remodelling enzyme from Equisetum (horsetails) and charophytic algae. Establish J. 55, 240–252.
Pubmed Abstract | Pubmed Total Text | CrossRef Total Text
Geisler-Lee, J., Geisler, Chiliad., Coutinho, P. M., Segerman, B., Nishikubo, N., Takahashi, J., Aspeborg, H., Djerbi, S., Master, E., Andersson-Gunneras, S., Sundberg, B., Karpinski, S., Teeri, T. T., Kleczkowski, L. A., Henrissat, B., and Mellerowicz, E. J. (2006). Poplar carbohydrate-active enzymes. Gene identification and expression analyses. Plant Physiol. 140, 946–962.
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Goubet, F., Jackson, P., Deery, K. J., and Dupree, P. (2002). Polysaccharide analysis using carbohydrate gel electrophoresis: a method to study constitute cell wall polysaccharides and polysaccharide hydrolases. Anal. Biochem. 300, 53–68.
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Hansen, S. F., Harholt, J., Oikawa, A., and Scheller, H. V. (2012). Plant glycosyltransferases beyond CAZy: a perspective on DUF families. Front. Establish Sci. 3:59. doi: 10.3389/fpls.2012.00059
Pubmed Abstract | Pubmed Total Text | CrossRef Total Text
Harholt, J., Sørensen, I., Fangel, J. U., Roberts, A., Willats, W. M. T., Scheller, H. V., Bent Larsen Petersen, B. Fifty., Banks, J. A., and Ulvskov, P. (2012). The glycosyltransferase repertoire of the spikemoss Selaginella moellendorffii and a comparative study of the cell wall structure. PLoS ONE 7, e35846. doi:10.1371/journal.pone.0035846
Pubmed Abstruse | Pubmed Full Text | CrossRef Full Text
Hepler, P. K., Fosket, D. E., and Newcomb, Eastward. H. (1970). Lignification during secondary wall formation in Coleus: an electron microscopy report. Am. J. Bot. 57, 85–96.
CrossRef Full Text
Hervé, C., Rogowski, A., Gilbert, H. J., and Knox, J. P. (2009). Enzymatic treatments reveal differential capacities for xylan recognition and degradation in primary and secondary plant cell walls. Found J. 58, 413–422.
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Iraki, N. G., Bressan, R. A., Hasegawa, P. Thou., and Carpita, N. C. (1989a). Alteration of the physical and chemical construction of the master cell wall of growth-limited plant cells adjusted to osmotic stress. Establish Physiol. 91, 39–47.
Pubmed Abstruse | Pubmed Total Text | CrossRef Total Text
Iraki, Due north. Chiliad., Singh, North., Bressan, R. A., and Carpita, N. C. (1989b). Cell walls of tobacco cells and changes in limerick associated with reduced growth upon adaptation to water and saline stress. Plant Physiol. 91, 48–53.
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Iraki, N. K., Bressan, R. A., and Carpita, Northward. C. (1989c). Extracellular polysaccharides and proteins of tobacco cell cultures and changes in composition associated with growth-limiting adaptation to water and saline stress. Plant Physiol. 91, 54–61.
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Kenrick, P., and Crane, P. R. (1997). The origin and early evolution of plants on land. Nature 389, 33–39.
CrossRef Full Text
Lerouxel, O., Choo, T. S., Seveno, M., Usadel, B., Faye, L., Lerouge, P., and Pauly, M. (2002). Rapid structural phenotyping of plant cell wall mutants by enzymatic oligosaccharide fingerprinting. Plant Physiol. 130, 1754–1763.
Pubmed Abstract | Pubmed Full Text | CrossRef Total Text
Mackie, West., and Sto, R. D. P. R. (1968). The occurrence of mannan microfibrils in the green algae Codium fragile and Acetabularia crenulata. Planta 253, 249–253.
CrossRef Total Text
Marcus, Due south. E., Blake, A. W., Benians, T. A. South., Lee, K. J. D., Poyser, C., Donaldson, L., Leroux, O., Rogowski, A., Petersen, H. L., Boraston, A., Gilbert, H. J., Willats, W. G. T., and Knox, J. P. (2010). Restricted admission of proteins to mannan polysaccharides in intact plant cell walls. Constitute J. 64, 191–203.
Pubmed Abstract | Pubmed Total Text | CrossRef Full Text
Matsunaga, T., Ishii, T., Matsumoto, S., Higuchi, M., Darvill, A., Albersheim, P., and O'Neill, M. A. (2004). Occurrence of the principal prison cell wall polysaccharide rhamnogalacturonan Two in pteridophytes, lycophytes, and bryophytes: implications for the evolution of vascular plants. Plant Physiol. 134, 339–351.
Pubmed Abstruse | Pubmed Full Text | CrossRef Full Text
Mitchell, R. A., Dupree, P., and Shewry, P. R. (2007). A novel bioinformatics arroyo identifies candidate genes for the synthesis and feruloylation of arabinoxylan. Establish Physiol. 144, 43–53.
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Moller, I., Sørensen, I., Bernal, A. J., Blaukopf, C., Lee, K., Öbro, J., Pettolino, F., Roberts, A., Mikkelsen, J. D., Knox, J. P., Bacic, A., and Willats, Due west. G. T. (2007). High-throughput mapping of cell-wall polymers inside and between plants using novel microarrays. Plant J. l, 1118–1128.
Pubmed Abstract | Pubmed Total Text | CrossRef Full Text
Mouille, G., Robin, S., Lecomte, G., Pagant, Southward., and Höfte, H. (2003). Nomenclature and identification of Arabidopsis prison cell wall mutants using Fourier-Transform InfraRed (FT-IR) microspectroscopy. Establish J. 35, 393–404.
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Niklas, Grand. J. (2004). The cell walls that bind the tree of life. BioScience 54, 831–841.
CrossRef Full Text
Park, S., Szumlanski, A. 50., Gu, F., Guo, F., and Nielsen, E. (2011). A office for CSLD3 during cell-wall synthesis in apical plasma membranes of tip-growing root-hair cells. Nat. Cell. Biol. 13, 973– 980.
Pubmed Abstruse | Pubmed Total Text | CrossRef Total Text
Pattathil, S., Avci, U., Baldwin, D., Swennes, A. K., McGill, J. A., Popper, Z., Bootten, T., Albert, A., Davis, R. H., Chennareddy, C., Dong, R., O'Shea, B., Rossi, R., Leoff, C., Freshour, M., Narra, R., O'Neil, One thousand., York, West. S., and Hahn, M. G. (2010). A comprehensive toolkit of plant cell wall glycan-directed monoclonal antibodies. Plant Physiol. 153, 514–525.
Pubmed Abstruse | Pubmed Full Text | CrossRef Full Text
Popper, Z. A., Michel, G., Hervé, C., Domozych, D. Southward., Willats, Westward. M., Tuohy, Thousand. G., Kloareg, B., and Stengel, D. B. (2011). Evolution and diversity of found cell walls: from algae to flowering plants. Annu. Rev. Establish. Biol. 62, 567–590.
Pubmed Abstract | Pubmed Total Text | CrossRef Full Text
Qu, Y., Egelund, J., Gilson, P. R., Houghton, F., Gleeson, P. A., Schultz, C. J., and Bacic, A. (2004). Identification of a novel group of putative Arabidopsis thaliana β-(1,three)-galactosyltransferases. Plant Mol. Biol. 68, 43–59.
Pubmed Abstruse | Pubmed Total Text | CrossRef Full Text
Scherp, P., Grotha, R., and Kutschera, U. (2001). Occurrence and phylogenetic significance of cytokinesis-related callose in light-green algae, bryophytes, ferns and seed plants. Found Jail cell Reports 20, 143–149.
CrossRef Full Text
Sørensen, I., Pettolino, F. A, Bacic, A., Ralph, J., Lu, F., O'Neill, Chiliad. A, Fei, Z., Rose, J. Thou. C., Domozych, D. Southward., and Willats, W. G. T. (2011). The charophycean green algae provide insights into the early on origins of plant cell walls. Plant J. 68, 201–211.
Pubmed Abstract | Pubmed Full Text | CrossRef Total Text
Spatz, H.-C., Rowe, North., Speck, T., and Daviero, V. (1998). Biomechanics of hollow stemmed Sphenopsids: Two. Calamites – to have or not to take secondary xylem. Rev. Palaeobot. Palynol. 102, 63–77.
CrossRef Total Text
Timme, R. E., and Delwiche, C. F. (2010). Uncovring the evolutionary origin of plant molecular processes: comparison of Coleochaete (Coleochaetales) and Spirogyra (Zygnematales) transcriptomes. BMC Institute Biol. 10, 96. doi: ten.1186/1471-2229-10-96
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Tsekos, I. (1999). The sites of cellulose synthesis in algae: diverseness and development of cellulose-synthesizing enzyme complexes. J. Phycol. 35, 635–655.
CrossRef Full Text
Velasquez, S. Chiliad., Ricardi, M. M., Dorosz, J. One thousand., Fernandez, P. V., Nadra, A. D., Pol-Fachin, L., Egelund, J., Gille, Due south., Harholt, J., Ciancia, M., Verli, H., Pauly, M., Bacic, A., Olsen, C. E., Ulvskov, P., Petersen, B. L, Somerville, C., Iusem, Due north. D., and Estevez, J. K. (2011). O-Glycosylated cell wall proteins are essential in root pilus growth. Science 332, 1401–1403.
Pubmed Abstract | Pubmed Full Text | CrossRef Full Text
Verhertbruggen, Y., Marcus, S. Due east., Haeger, A., Verhoef, R., Schols, H. A., McCleary, B. V., McKee, L., Gilbert, H. J., and Knox, J. P. (2009). Developmental complexity of arabinan polysaccharides and their processing in establish jail cell walls. Institute J. 59, 413–425.
Pubmed Abstruse | Pubmed Full Text | CrossRef Full Text
Vogel, J. P., Garvin, D. F., Mockler, T. C., Schmutz, J., Rokhsar, D., Bevan, M. W., Barry, K., Lucas, Due south., Harmon-Smith, One thousand., Lail, 1000., Tice, H., Schmutz, J., Grimwood, J., McKenzie, N., Bevan, M. Due west., Huo, N., Gu, Y. Q., Lazo, Chiliad. R., Anderson, O. D., Vogel, J. P., Yous, F. M., Luo, M. C., Dvorak, J., Wright, J., Febrer, M., Bevan, Grand. W., Idziak, D., Hasterok, R., Garvin, D. F., Lindquist, Due east., Wang, M., Play tricks, Due south. E., Priest, H. D., Filichkin, S. A., Givan, S. A., Bryant, D. W., Chang, J. H., Mockler, T. C., Wu, H., Wu, Due west., Hsia, A. P., Schnable, P. Due south., Kalyanaraman, A., Barbazuk, B., Michael, T. P., Hazen, S. P., Bragg, J. Due north., Laudencia-Chingcuanco, D., Vogel, J. P., Garvin, D. F., Weng, Y., McKenzie, N., Bevan, Yard. Westward., Haberer, G., Spannagl, Thou., Mayer, K., Rattei, T., Mitros, T., Rokhsar, D., Lee, S. J., Rose, J. K., Mueller, L. A., York, T. L., Wicker, T., Buchmann, J. P., Tanskanen, J., Schulman, A. H., Gundlach, H., Wright, J., Bevan, M., de Oliveira, A. C., Maia Lda, C., Belknap, W., Gu, Y. Q., Jiang, N., Lai. J., Zhu, Fifty., Ma, J., Sunday, C., Pritham, Due east., Salse, J., Murat, F., Abrouk, Grand., Haberer. G., Spannagl. One thousand., Mayer, M., Bruggmann, R., Messing, J., Y'all, F. Thousand., Luo, M. C., Dvorak, J., Fahlgren, North., Fox, Due south. E., Sullivan, C. Thou., Mockler, T. C., Carrington, J. C., Chapman, E. J., May, One thousand. D., Zhai, J., Ganssmann, 1000., Gurazada, S. Thou., German, M., Meyers, B. C., Green, P. J., Bragg, J. Northward., Tyler, Fifty., Wu, J., Gu, Y. Q., Lazo, G. R., Laudencia-Chingcuanco, D., Thomson, J., Vogel, J. P., Hazen, South. P., Chen, S., Scheller, H. 5., Harholt, J., Ulvskov, P., Play a joke on, Southward. E., Filichkin, Due south. A., Fahlgren, Due north., Kimbrel, J. A., Chang, J. H., Sullivan, C. M., Chapman, E. J., Carrington, J. C., Mockler, T. C., Bartley, Fifty. E., Cao, P., Jung, K. H., Sharma, M. K., Vega-Sanchez, 1000., Ronald, P., Dardick, C. D., De Bodt, S., Verelst, W., Inzé, D., Heese, M., Schnittger, A., Yang, X., Kalluri, U. C., Tuskan, K. A., Hua, Z., Vierstra, R. D., Garvin, D. F., Cui, Y., Ouyang, S., Sun, Q., Liu, Z., Yilmaz, A., Grotewold, East., Sibout, R., Hematy, Thou., Mouille, Thousand., Höfte, H., Michael, T., Pelloux, J., O'Connor, D., Schnable, J., Rowe, South., Harmon, F., Cass, C. L., Sedbrook, J. C., Byrne, M. Due east., Walsh, S., Higgins, J., Bevan, M., Li, P., Brutnell, T., Unver, T., Budak, H., Belcram, H., Charles, M., Chalhoub, B., and Baxter, I. (2010). Genome sequencing and analysis of the model grass Brachypodium distachyon. Nature 463, 763–768.
Pubmed Abstruse | Pubmed Total Text | CrossRef Full Text
Vreeland, 5., Slomich, One thousand., and Laetsch, W. Yard. (1984). Monoclonal antibodies as molecular probes for prison cell wall antigens of the chocolate-brown alga Fucus. Planta 163, 506–517.
CrossRef Full Text
Willats, Due west. M. T., Orfila, C., Limberg, G., Buchholt, H. C., van Alebeek, Chiliad.-J. W. M., Voragen, A. 1000. J., Marcus, S. Due east., Christensen, T. M. I. E., Mikkelsen, J. D., Murray, B. S., and Knox, J. P. (2001). Modulation of the degree and pattern of methyl-esterification of pectic homogalacturonan in found cell walls: implications for pectin methyl esterase action, matrix properties and cell adhesion. J. Biol. Chem. 276, 19404–19413.
Pubmed Abstruse | Pubmed Full Text | CrossRef Full Text
Wodniok, S., Brinkmann, H., Glöckner, Thou., Heidel, A. J., Philippe, H., Melkonian, 1000., and Becker, B. (2011). Origin of land plants: do conjugating green algae concord the central? BMC Evol. Biol. 11, 104. doi: 10.1186/1471-2148-xi-104
Pubmed Abstract | Pubmed Total Text | CrossRef Full Text
Yin, Y., Chen, H., Hahn, Thousand. Thou., Mohnen, D., and Xu, Y. (2010). Evolution and function of the plant cell wall synthesis-related glycosyltransferase family 8. Plant Physiol. 153, 1729–1746.
Pubmed Abstract | Pubmed Total Text | CrossRef Total Text
Source: https://www.frontiersin.org/articles/10.3389/fpls.2012.00152/full
0 Response to "What Benefit Does Having a Cell Wall Made of Cellulose Provide to Land Plants"
Post a Comment